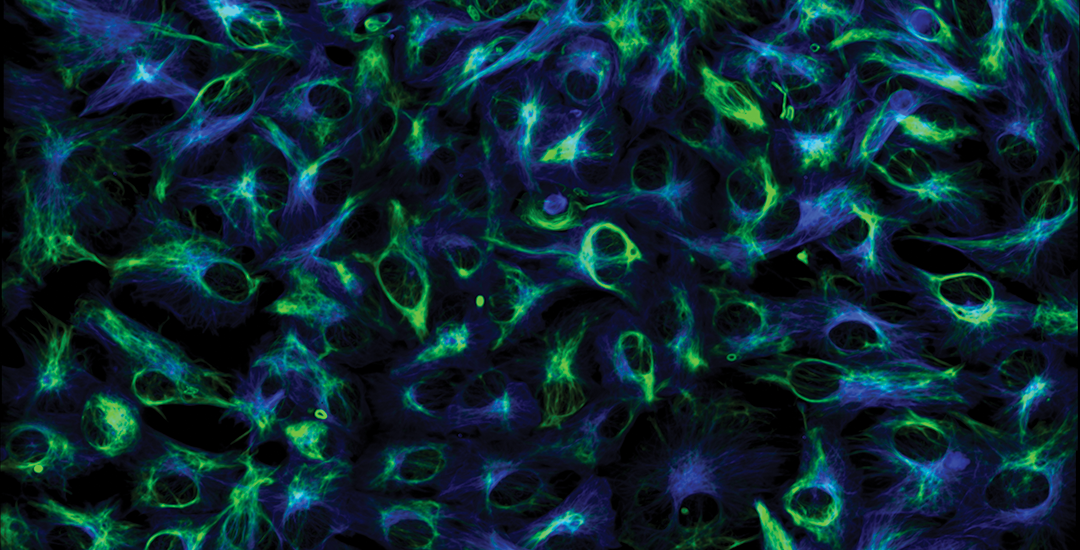
The Best Solution for Live Cell Imaging Where Both Optical Sectioning and Cell Viability are Critically Important

Yokogawa spinning disk confocals incorporate two Nipkow disks, one with pinholes for sharp confocal imaging and the other with microlens pinholes to illuminate with many more photons than otherwise would pass through the disk. The result is a great improvement in signal while maintaining strong confocality with discrete optical sectioning. Unlike laser scanning confocals which scan a single point of light across a field, a spinning disk confocal scans hundreds of points of light across the same field simultaneously resulting in much faster imaging. This significant speed difference combined with the superior sensitivity of back-thinned sCMOS cameras make spinning disk confocal a must-have technology for live cell imaging labs.
Yokogawa CSU-W1 for High Speed High Resolution Live Cell Imaging
| Pinhole Diameter | 25µm disk and 50µm disk |
|---|---|
| Number of Disks | Up to two with motorized switching |
| Number of Cameras | Up to two with motorized switching |
| Filter Wheel Positions | Up to ten per camera with motorized switching |
| Disk Bypass for Widefield Imaging | Standard |
| Acquisition Speed | 200 FPS |
| Field of View | 17mm x 16mm |
| Near IR Excitation | Up to 785nm |
| Motorization | Disk Exchange, Variable Aperture, Camera Selection & Camera Magnification |
| Options | Split-View Imaging, NIR Imaging, Illumination Field Flattening & Super-Resolution |

Yokogawa CSU-W1 Super-Resolution by Optical Re-Assignment (SoRa)
CSU-W1 SoRa is an easy-to-use super-resolution microscopy solution utilizing a dual Nipkow disk assembly with microlenses on both the illuminating and imaging disks. SoRa images have a 1.4x resolution improvement and deconvolved SoRa images have a 2x resolution improvement compared to standard spinning disk data. With a maximum speed of 200fps, low phototoxicity and no limitation on dyes or fluors, SoRa is ideal for super-resolution live cell imaging. SoRa is also available as an upgrade to existing CSU-W1 systems.
| Imaging Speed | 200 fps |
|---|---|
| Wavelength Range | 405nm to 640nm Excitation 420nm to 680nm emission |
| XY Resolution | 150nm 120nm with deconvolution |
| Z Resolution | 320nm 300nm with deconvolution |
| Field of View 63x Objective and 4x Relay Magnification |
67µm x 63µm |
| Field of View 100x Objective and 2.8x Relay Magnification |
61µm x 57µm |

Standard

Standard Deconvolved

SoRa

SoRa Deconvolved

Argolight test slide (520nm emission) imaged with Plan-Apochromat 100x/1.46NA objective and 2.8x magnification. SoRa achieves a resolution of 150nm, improved to 120nm with deconvolution.
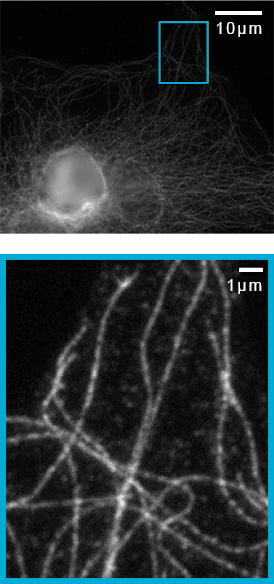
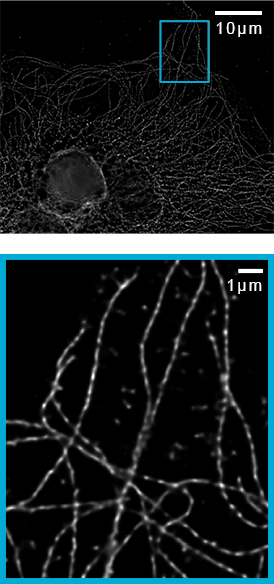
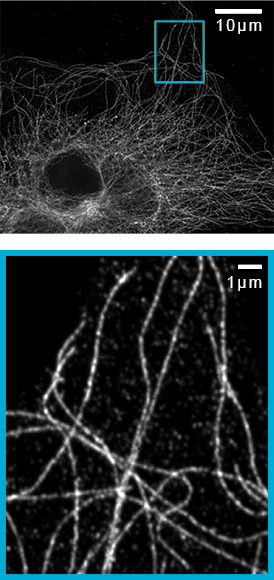
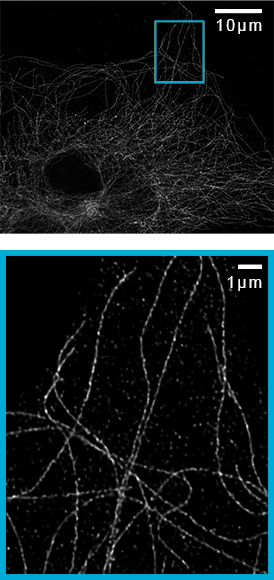
Imaging of microtubules in fixed bovine pulmonary artery endothelial cells. Azuma, T. and Kei, T. (2015) Super-resolution spinning-disk confocal microscopy using optical photon reassignment. Opt Express. Jun 1;23(11):15003-11. doi: 10.1364/OE.23.015003.
Uniformizer | Illumination Field Uniformity
For exceptionally even illumination across the entire field, Uniformizer conditions the gaussian beam from the illumination fiber optic to distribute light evenly across the field. Using a set of microlens arrays, Uniformizer flattens the field to as little as 1% variance and boosts overall intensity up to 50%.

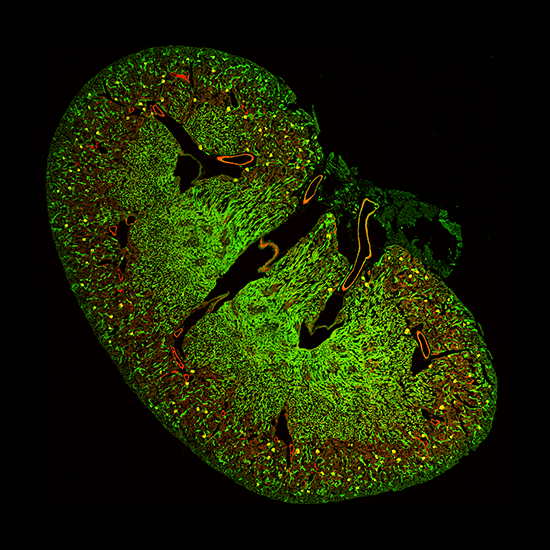
Standard Montage
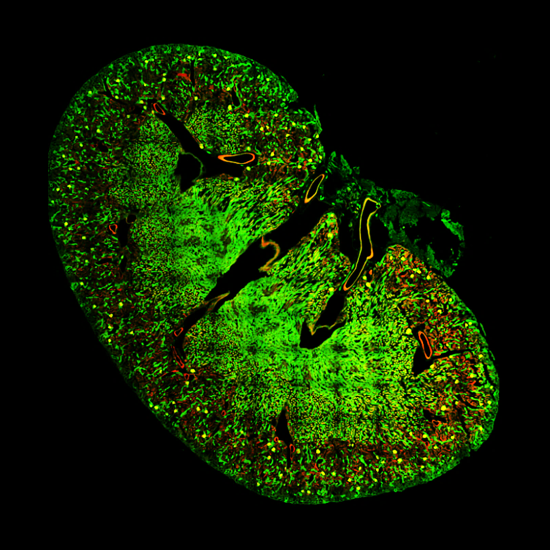
With Uniformizer
![100x Montage - Position 1 [25] Montage.Project Maximum Z](https://www.intelligent-imaging.com/wp-content/uploads/2022/03/100x-Montage-Position-1-25-Montage.Project-Maximum-Z.png)
Prostate tissue section with nuclei in blue and vasculature in orange. Imaged with Marianas SDC and Uniformizer, 100x/1.46NA objective. Image shown is 650µm across montaged with 2 x 6 fields of view.
Yokogawa CSU-X1
| Pinhole Diameter | 50µm disk |
|---|---|
| Number of Disks | One |
| Disk Bypass | With 3i bypass |
| Acquisition Speed | 2000 FPS |
| Effective Field of View | 10mm x 7mm |
| Near IR Excitation | Up to 640nm |
| Motorization Options | Camera Port Selection, Disk Dichroic and Emission Filter Change |

Spinning Disk Confocal Data Sets
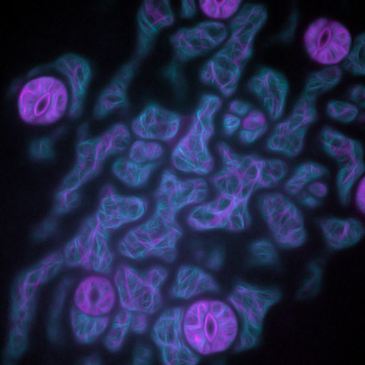
PLANT BIOLOGY
Actin (magenta) and microtubule cytoskeleton (cyan) in Arabidopsis thaliana cotyledons. Courtesy of Isabella Østerlund and Dr. Felix Ruhnow, Dr. Staffan Persson group at University of Copenhagen.
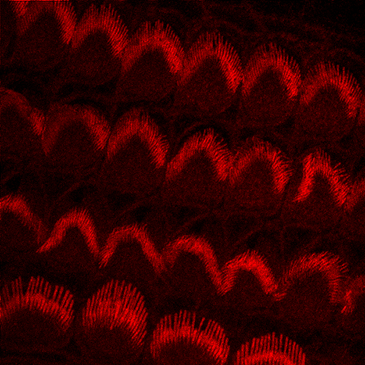
Ex Vivo
Murine hair follicles. Courtesy of Dr. Anthony Peng, Massachusetts Institute of Technology.
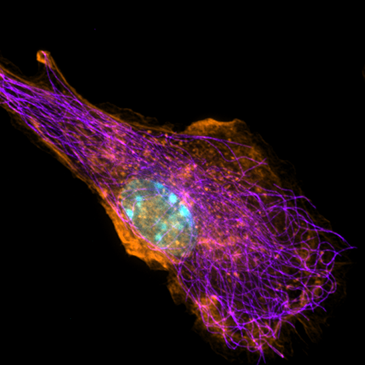
Intracellular Imaging
Macrophage labeled for nucleus (blue), microtubules (purple), and actin (orange).
![100x Montage 2.8x - Position 1 [49] Montage.Project Maximum Z](https://www.intelligent-imaging.com/wp-content/uploads/2022/04/100x-Montage-2.8x-Position-1-49-Montage.Project-Maximum-Z.png)
Cancer Research
Prostate tissue section labeled for nuclei (blue) and blood vessels (orange).
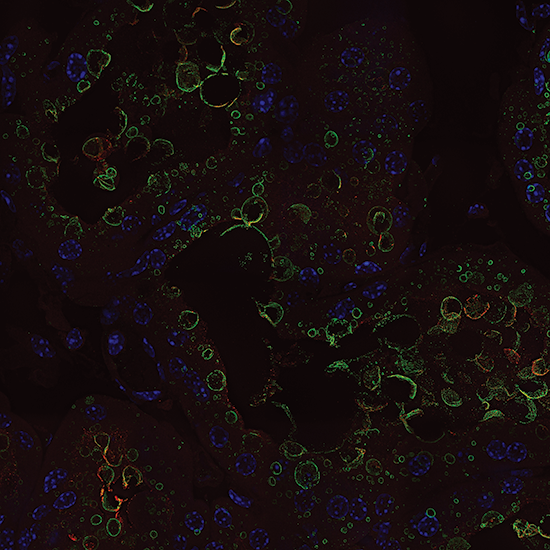
Reproductive Medicine
Lipid docks in lactating mammary epithelial cells. 20x montage. Courtesy of Dr. Jenifer Monks, CU Anschutz Medical Campus.
Developmental Biology
Drosophila pupa wing disc expressing E-cadherin GFP.
Developmental Biology
Zebrafish tail labeled for actin.