Total Internal Reflection Fluorescence and Photomanipulation Module
Vector3 is a motorized, spinning TIRF illuminator with three key imaging modalities: TIRF, photomanipulation and widefield epifluorescence. Intelligent beam steering and optical design allow for all three imaging modes to be combined in one compact device. Vector3 offers an expansive TIRF field of view (FN20) designed for modern sCMOS cameras. A motorized scan lens corrects for sample height variation and ensures ideal TIRF illumination and photomanipulation spot size across the visible spectrum.
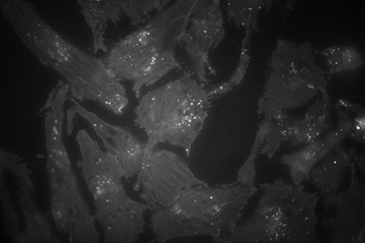
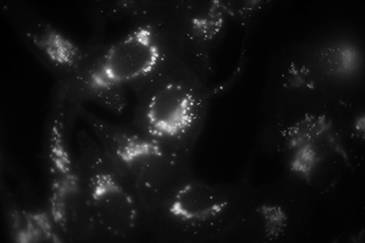
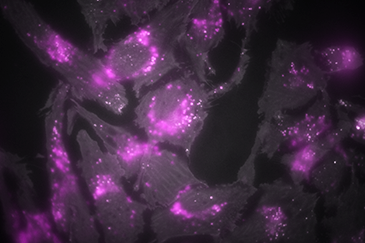
Comparison between TIRF (left), wide field episode-fluorescence (middle) and a composite of both (right). When in TIRF, only the bottom of the cells are visualized, leading to a large increase in signal to noise of the membrane and organelles interacting with the membrane.
All Optical Electrophysiology
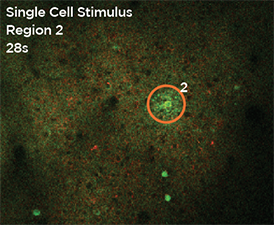
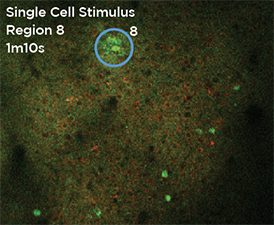
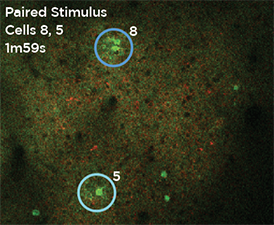
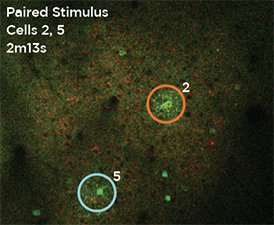
Nouveau Phasor is designed for 3D 2-photon stimulation of neurons in vivo. Four distinct modalities enable flexible optogenetics studies involving organisms from C. elegans and Drosophila to zebrafish, mice and larger mammalian species. Nouveau Phasor is able to simultaneously illuminate multiple regions in a 3D volume with the use of SLM-based computer-generated holography.
Scanning Photomanipulation
Vector2 is a diffraction-limited high speed X,Y scanner that accepts lasers from UV to IR. Vector allows interactive examination of living specimens with a rapid single-point seek time of 0.3ms and fast ROI scanning (1500 lines per second). Common uses of Vector2 include FRAP, photoactivation, photoablation and uncaging. mSwitcher enables simultaneous use with Spinning Disk Confocal for advanced multi-modal methods. Vector2 is also designed for high-speed scanning imaging techniques such as multiphoton imaging or laser scanning confocal microscopy
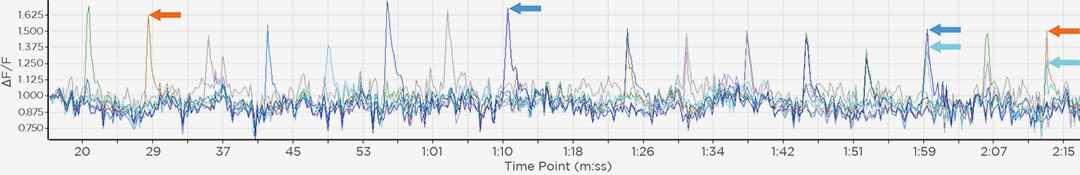
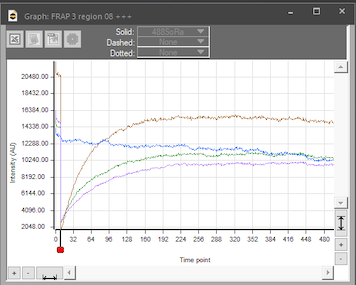
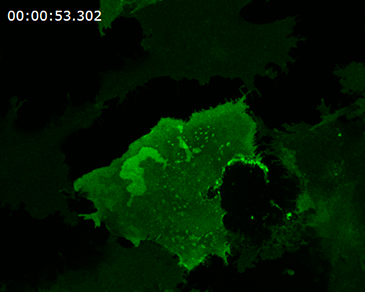
photobleaching
Mammalian cell with membrane labeled in green before bleaching and FRAP curve in SlideBook.
Laser Ablation System
Ablate! uses an attenuatable 355nm or 532nm pulse laser (>60µJ pulses at 200Hz) to inflict localized damage to a diffraction-limited spot in intracellular structures for precise laser ablation and laser wounding. Full software control with SlideBook features automated intensity control and allows users to define one or more regions of interest as single diffraction limited spots or as user-defined shapes.
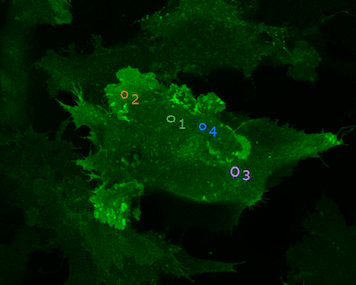
Photoablation
Mammalian cell with membrane labeled in green before (left) and after ablation (right)
Holographic Photomanipulation
Phasor 1-Photon uses a phase-only spatial light modulator (SLM) designed specifically for patterned and 3D point photomanipulation in optical microscopy. Phasor is compact and modular, allowing direct attachment to a microscope imaging port. Illumination is provided by LaserStack through a fiber optic coupling. Phasor is controlled by SlideBook software to manage region specification, hologram generation, experimentally synchronized optical path switching and excitation light gating. This state of the art control enables sophisticated experiment design incorporating pattern photomanipulation, including FRAP, photoactivation, photoconversion, and photostimulation.