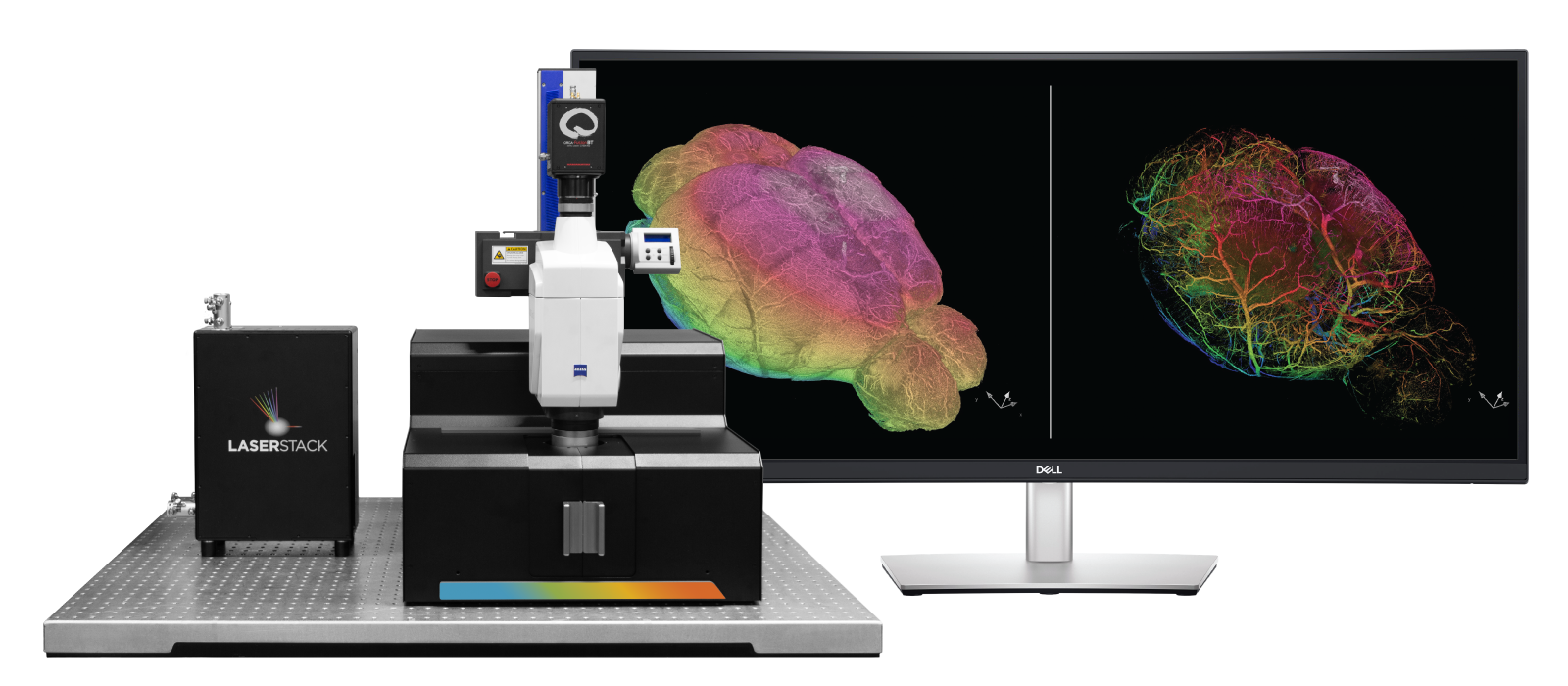
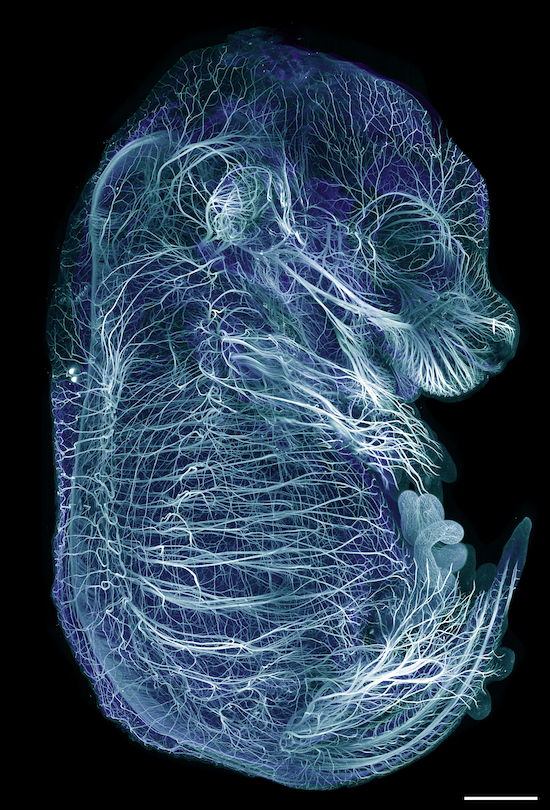
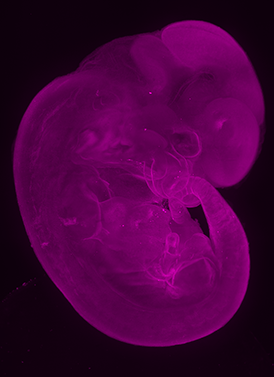
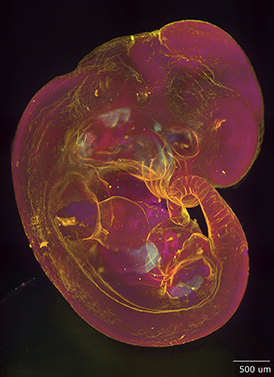
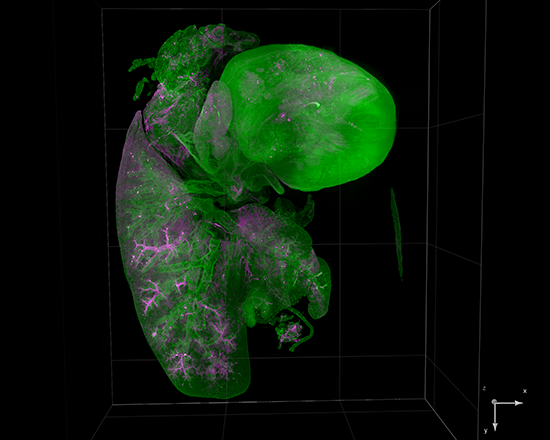
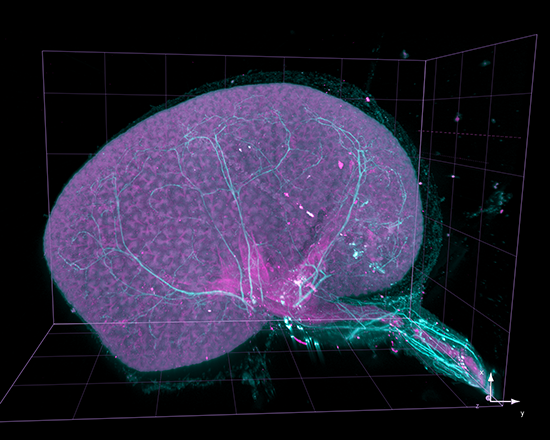
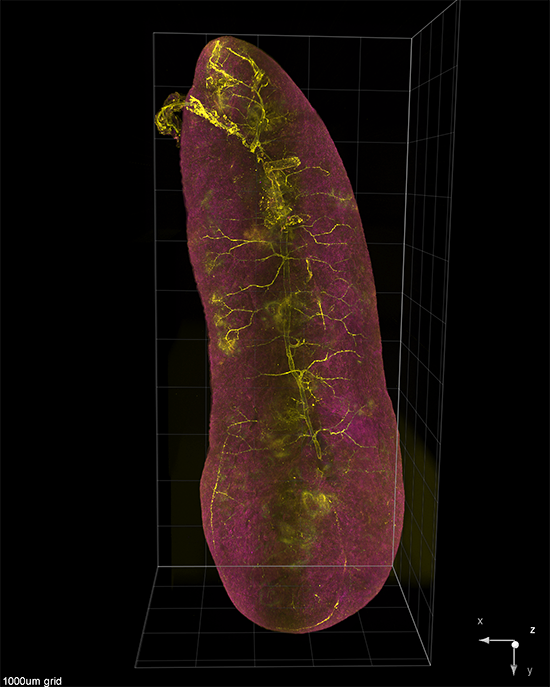
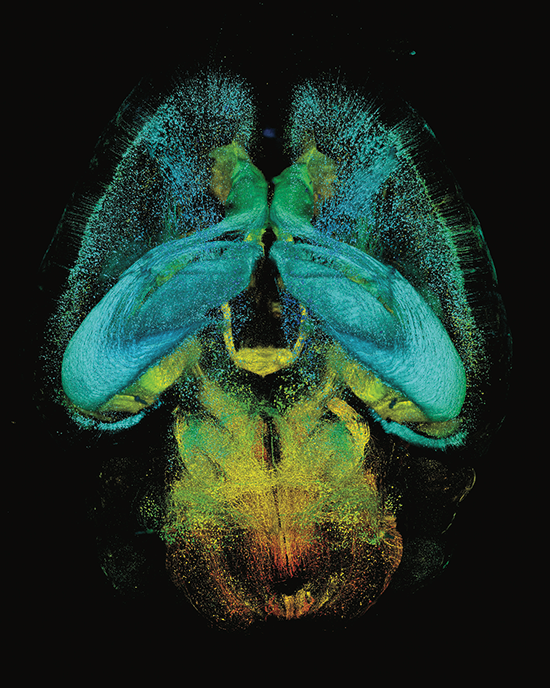
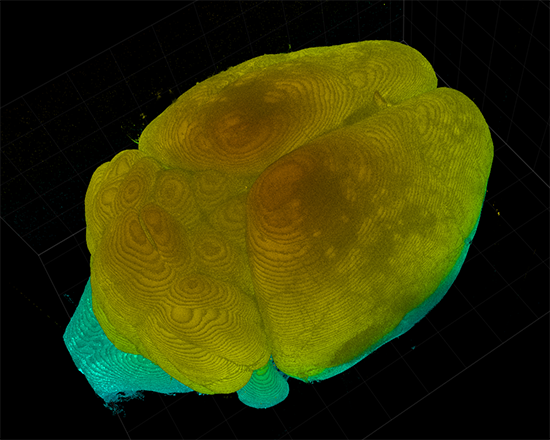
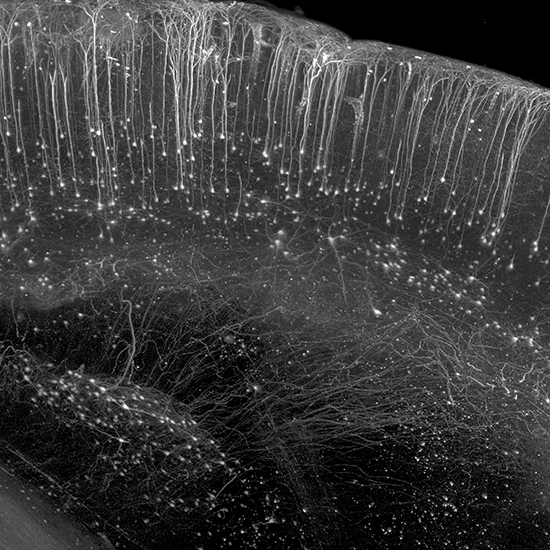
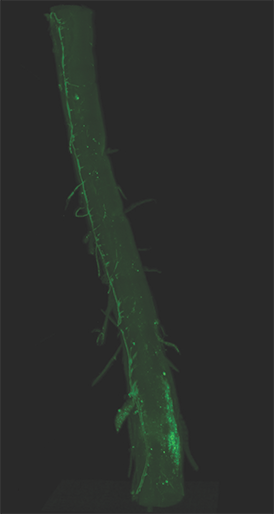
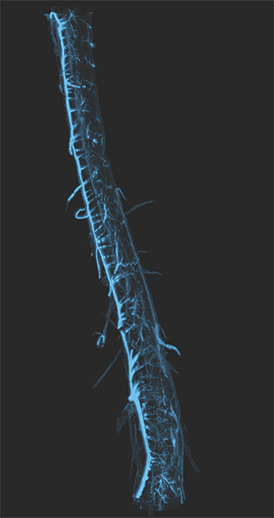
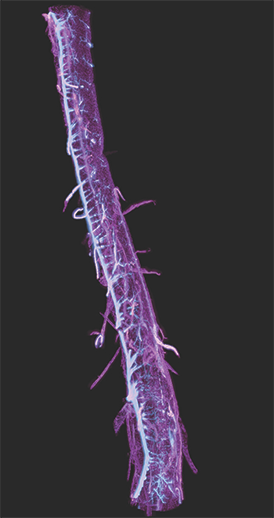
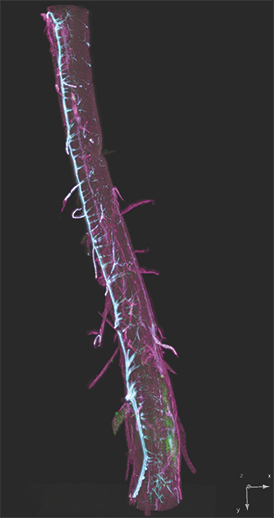
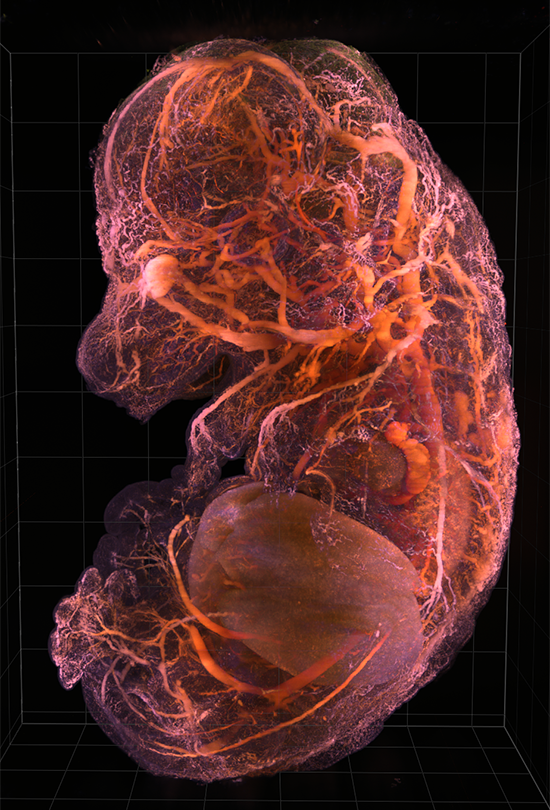
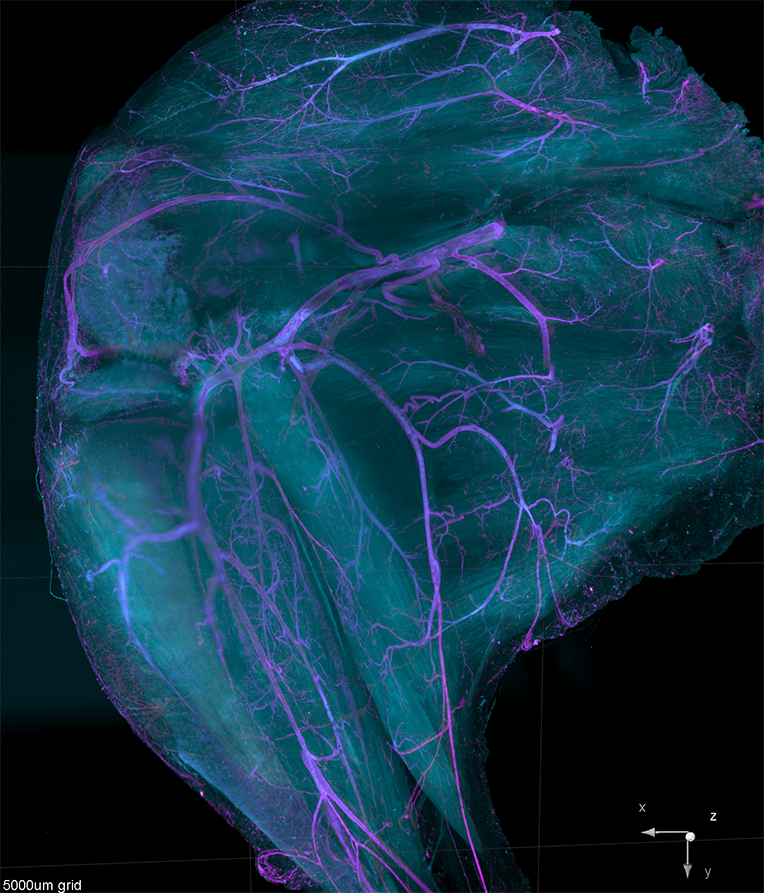
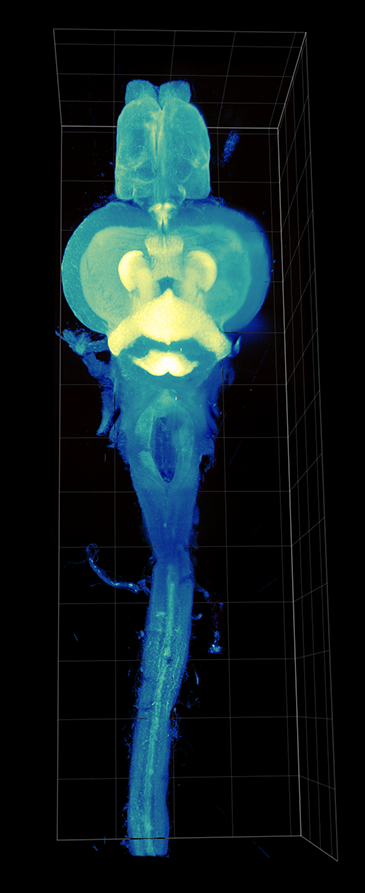
AxL CTLS Scanner
Proprietary dual-sided light sheet illumination optics
Compact 54 x 60cm footprint
Seamlessly switch imaging modes without changing optics - Axially swept light sheet mode & Ultrafast 3D prescan mode
Back-thinned sCMOS Camera
Multichannel Z Galvo
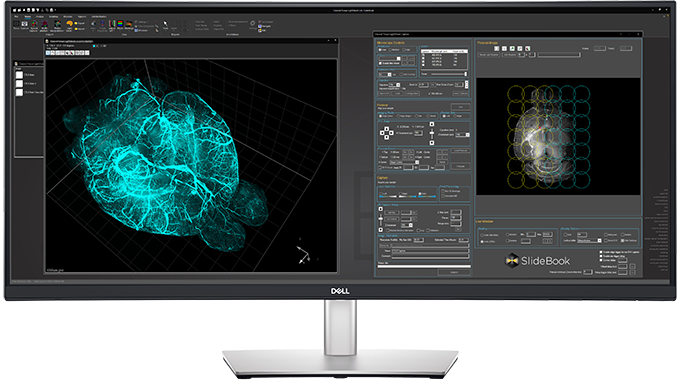
Automated Zoom Microscope



2304 x 2304 6.5µm pixels
95% quantum efficiency
Rolling shutter mode
High-speed axial chromatic focus adjustment
Motorized 16:1 zoom for optimal pixel sampling


Electrically Tunable Lens
Macro Fluorescence Objectives
LaserStack Laser Combiner
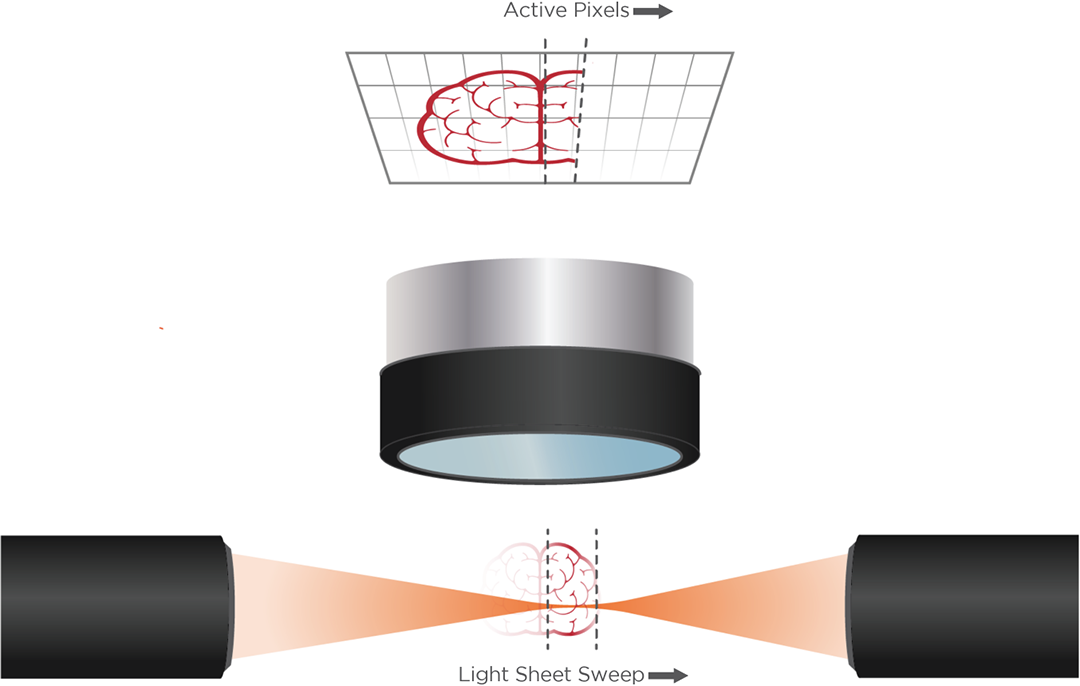
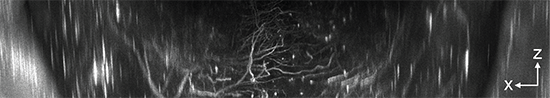
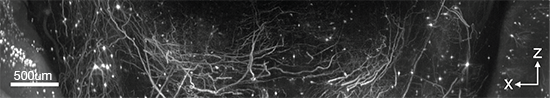
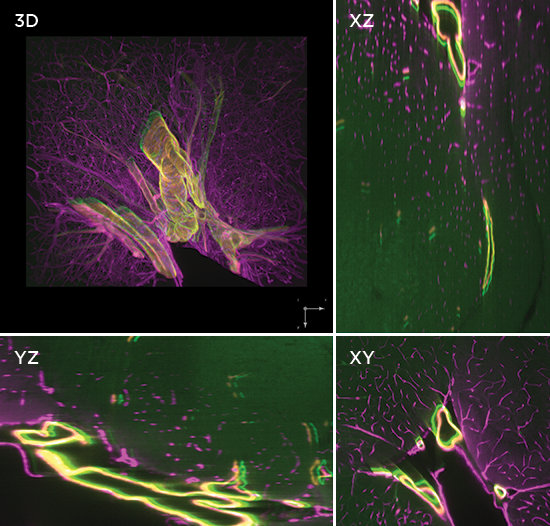
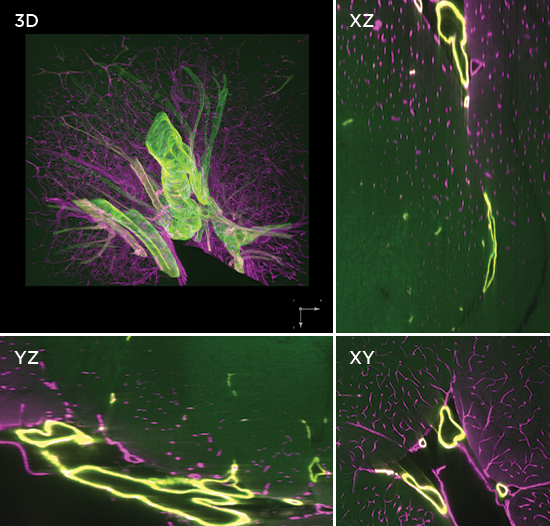
Axial light sheet sweep length of 10mm for large field of view (FOV)
1.0x/0.25NA, WD 56mm
1.5x/0.37NA, WD 30mm
2.3x/0.57NA, WD 10mm
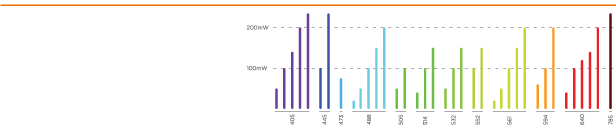
Modular Laser Combiner
Up to 8 lasers
Up to 300mW



Dynamic Galvo-Scanned Light Sheet
Custom Excitation Objectives
Chambers and Specimen Holders
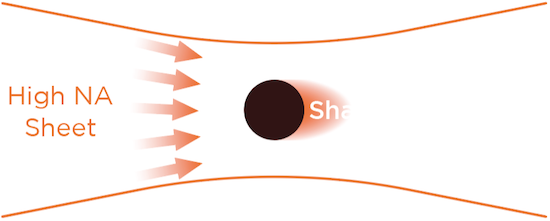
Thin and uniform sheet across a 10mm FOV
Adjustable FOV for faster imaging of smaller specimens
Spherical aberration correction for high-refractive-index immersion media
Flat excitation profile across 1mm to 10mm sheet width
0.14NA diffraction-limited light sheet
Sample chambers in 4 different sizes
Temperature controlled
Chemically-inert sample holders
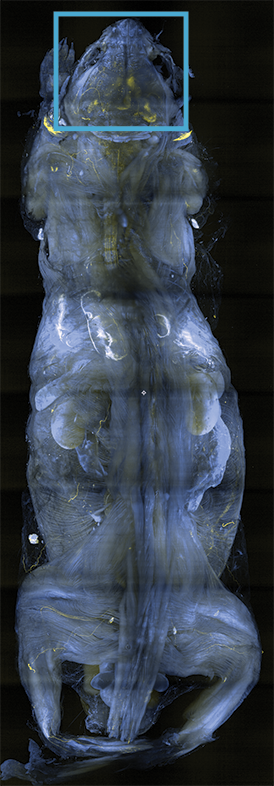
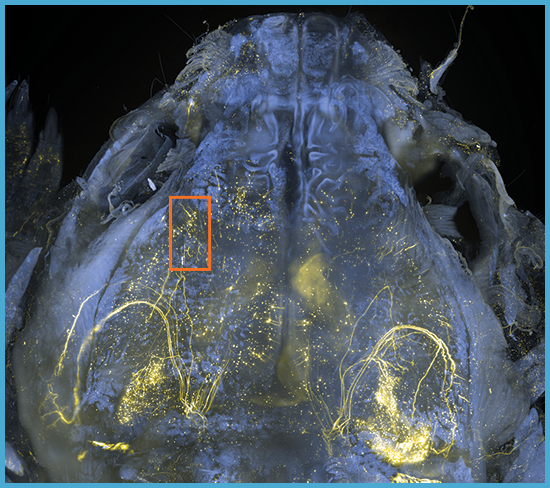
Clearing solutions ranging from 1.33-1.56 RI
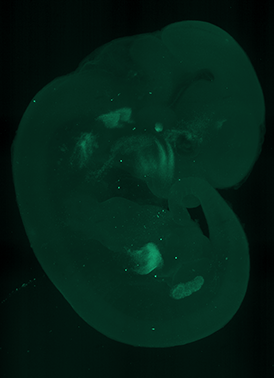
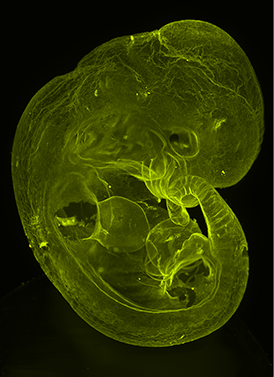
Multi-specimen holder for up to 10 different samples